
Everyone wants to be happy. However, according to psychologists, many of us think in certain “unhelpful” ways which can prevent us from being happy. The free website at the link below describes the concept of “Happy Thinking”, which is about using simple methods to change such ways of thinking. You are welcome to visit it and find happiness by changing how you think. The website is completely free.

What is a vapour ?
Inhalational anaesthetic agents need to be delivered to the lungs for them to work. Of course one cannot simply pour them into the lungs !

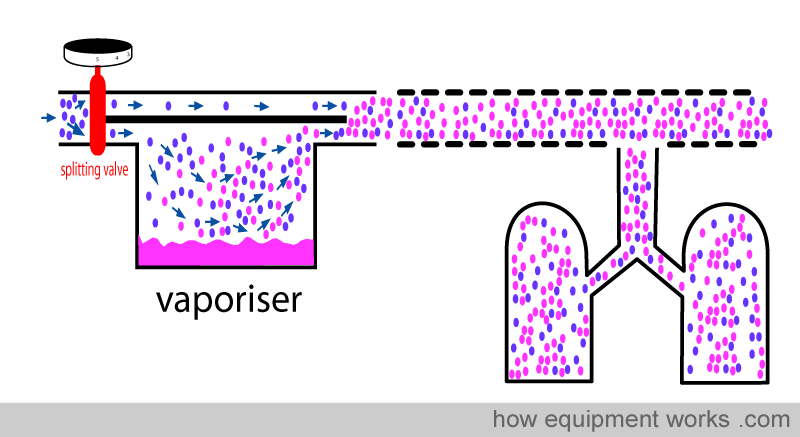
A much more acceptable way is to convert the liquid inhalational agent into a form that can be delivered by the inhalational route to the patient. The most convenient way is to deliver the “vapour “ form of the liquid inhalational agent. A device which converts liquid to vapour is called a vaporiser.
Before going onto discussing vaporisers, we need to first understand what a vapour is. And to understand what a vapour is, we need to know about something called critical temperature.
If you take a gas, and compress it really hard, the particles that compose it are brought ever so close to each other. As you keep compressing , the particles will at some point coalesce and convert the gas into liquid. However, if the gas is above a certain temperature, called a “critical temperature”, whatever amount of pressure you apply, that gas will not become a liquid. This temperature is called “critical temperature” and every gas has its particular critical temperature.
A gas that is currently below its critical temperature is called a “vapour “. If compressed with enough pressure, it will condense into a liquid.
A gas that is currently above its critical temperature remains a gas. However hard you compress it, it will not condense into a liquid.
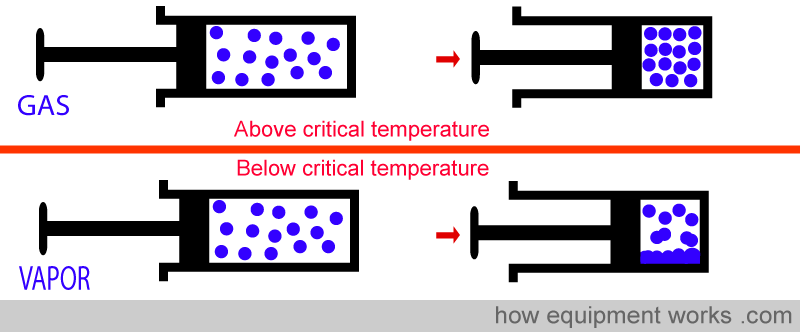
Let us take isoflurane as a example. The critical temperature of isoflurane is about 200 degrees centigrade. Therefore, at room temperature (e.g. 21 degrees centigrade), the gaseous phase of isoflurane would be called “isoflurane vapour “.
Now for a moment, let us imagine that you worked on the planet Venus. The surface temperature on Venus is about 500 degrees centigrade.

Now, because the “˜room temperature on Venus’ (500 C) is higher than the critical temperature of isoflurane (200 C), the gaseous phase of isoflurane would be called “isoflurane gas “.
Let us come back to Earth. If all this is confusing you, just remember, on Earth, at room temperature, all the gaseous forms of common anesthetic agents exist as vapours.
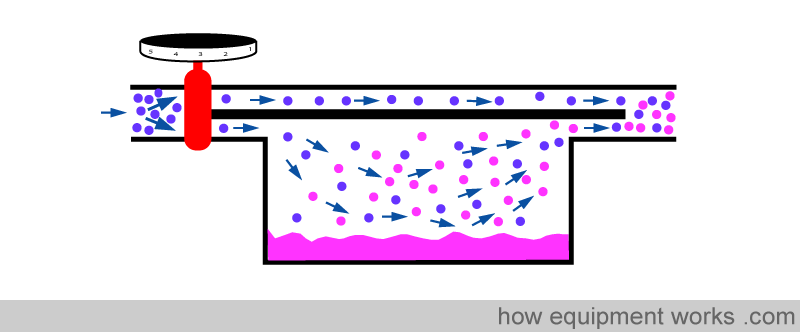
Basic Vaporiser
The purpose of a vaporiser is to add anaesthetic vapor into the fresh gas flow in a way that the output of the vaporiser delivers the set concentration of anaesthetic agent accurately.
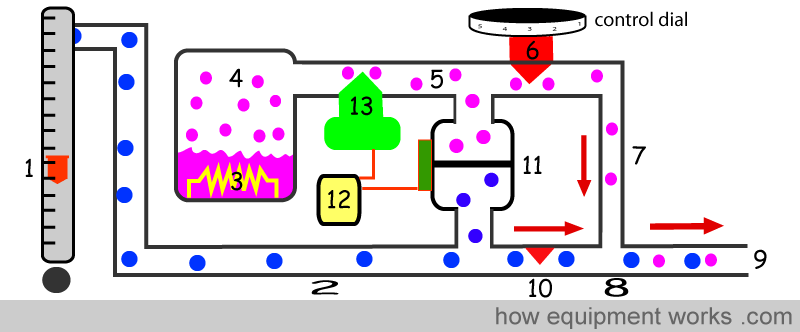
Fresh gas enters the inlet of the vaporiser and is divided into two flow pathways. The splitting valve, depending on the setting of the control dial, adjusts how much goes through each of the pathways. The fresh gas that is sent along the “by pass” pathway doesn’t come into contact with any vapor. On the other hand, the fresh gas that is sent to the vaporising chamber becomes fully saturated with vapor. At the exit end of the vaporiser, the by pass gas (vaporless) meets the chamber gas (fully saturated with vapor) and the two mix. The resultant output depends on how much of fresh gas went though each of the pathways.
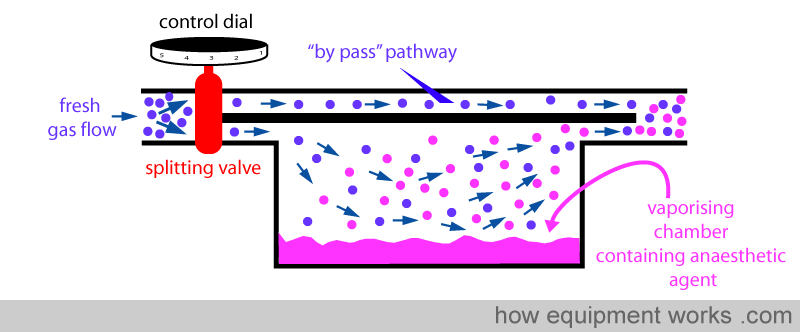
When you dial a high anaesthetic concentration requirement, the splitting valve sends more fresh gas via the vaporising chamber.
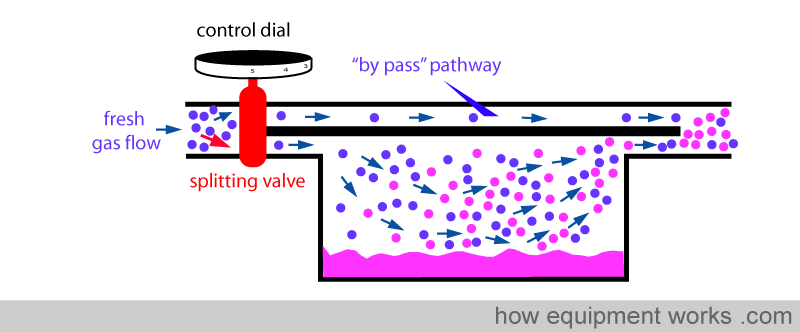
Similarly, when you dial a low anaesthetic concentration requirement, the splitting valve sends less fresh gas via the vaporising chamber.
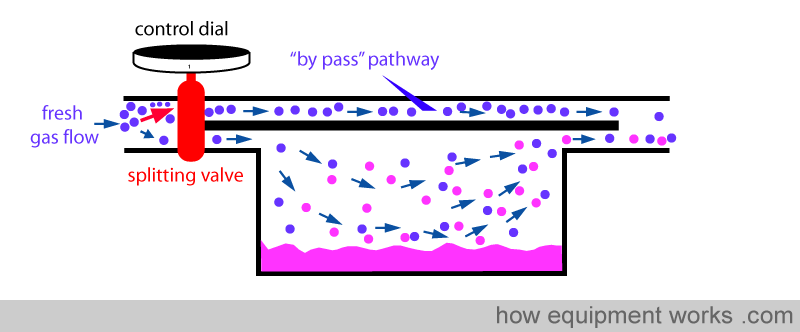
Finally, when you set the dial to zero to make vaporiser deliver no anaesthetic vapor, the splitting valve sends all the fresh gas via the by pass pathway and nothing through the vaporising chamber.
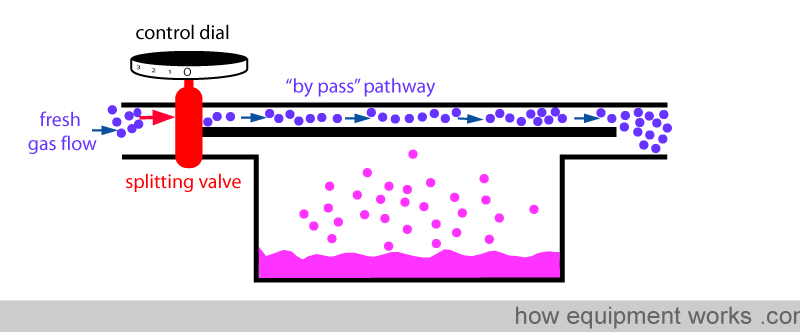
You have seen that the anaesthetic concentration that is output by the vaporiser is determined by the ratio of fresh gas flow that goes through the vaporising chamber and the fresh gas flow that goes through the bypass pathway. This ratio is called the ‘splitting ratio’.
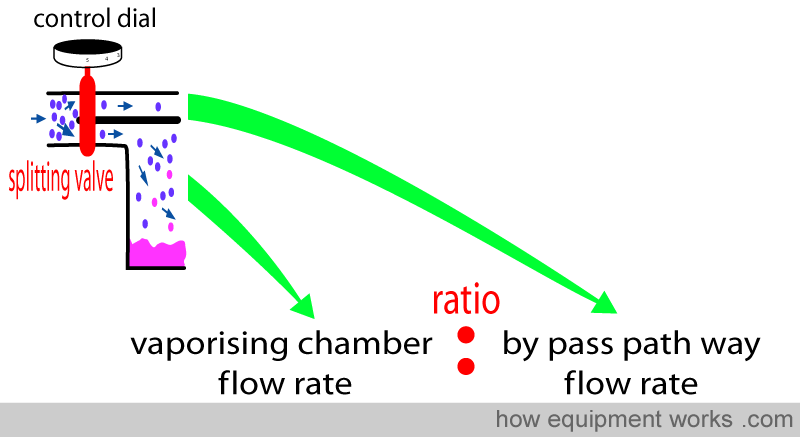
Problems of the Basic Design
The basic vaporiser discussed above has a very simple design. Unfortunately, this simple design has the following problems:
- High flows of fresh gas going through the whole vaporiser can affect its output.
- The temperature of the vaporiser drops with use and this can affect its output.
- Some ventilators transmit a “positive pressure” back into vaporiser which can affect its output.
Problem of High Flow
As discussed before, part of the fresh gas flow enters the vaporisation chamber and picks up vapor.
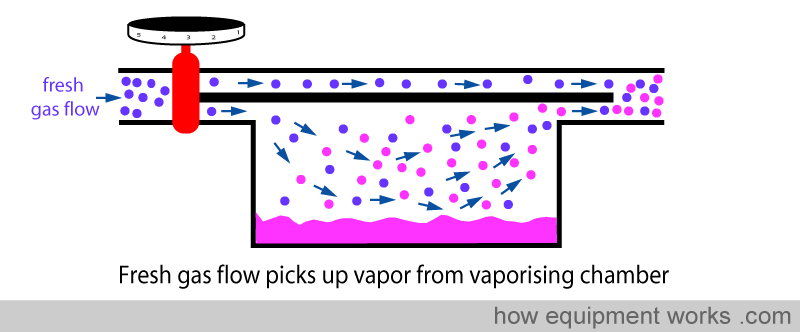
However, in the basic design, this vaporisation is not very efficient. If one uses a high fresh gas flow, the vaporisation process can’t keep up with so much gas arriving into the vaporisation chamber. The result is that, relative to the high flow of fresh gas flow, the amount of anaesthetic vaporised is inadequate. So this means that at high flows, the basic vaporiser delivers less anaesthetic concentration than is set on the dial.
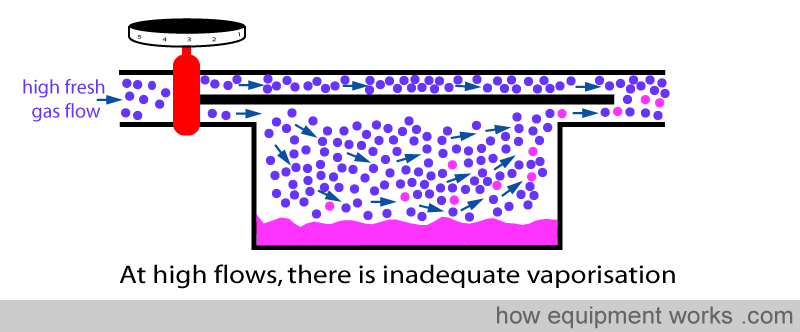
The solution employed by modern vaporisers to solve this problem is to make the vaporisation much more efficient by increasing the surface area of contact between the fresh gas and anesthetic agent. So even when there are high flows, the efficient vaporisation means that all gas going through the vaporisation chamber is fully saturated. Because of this ability to saturate fresh gas at all flow rates, the output concentration remains accurate to the setting on the dial over a wide range of flows. I.e. The output concentration is independent of flow.
One method that vaporisers use to increase the efficiency of vaporisation is to dip wicks into the anaesthetic agent. Due to capillary action, the anaesthetic agent rises into the wicks. This dramatically increases the surface area of anaesthetic agent exposed to the fresh gas entering the vaporisation chamber and thereby improves the efficiency of vaporisation.

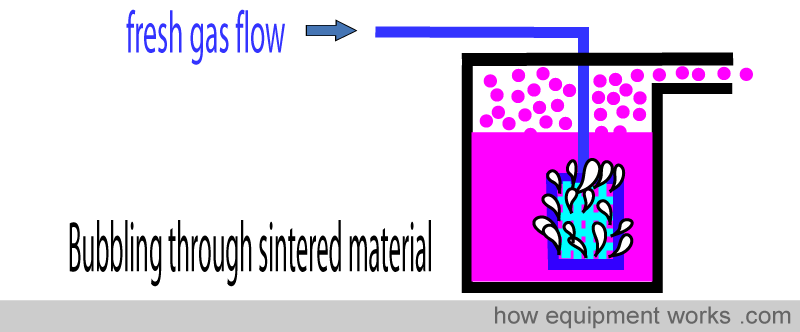
Certain vaporisers (e.g. “Copper Kettle”) use bubbles to increase the surface area for vaporisation. In these, some of the fresh gas flow is bubbled through a disk made out of a special material (sintered disk) that is very porous. The disk is submerged into the anaesthetic agent and when fresh gas is sent through it, a large number of tiny bubbles form. The tiny bubbles of fresh gas have a very large total surface and thus become fully saturated with vapor efficiently.
Problem of Temperature
For vaporisation to occur, the anaesthetic molecules have to “escape” from the liquid state and become vapor. This process reduces the ‘energy’ left in the remaining liquid.
As more and more molecules escape, more and more energy is lost from the liquid. The temperature of a liquid is a measurement of how much ‘heat energy’ the liquid has. Therefore, as the escaping molecules reduce the energy left in the liquid, the temperature of the liquid falls.
The falling temperature (lowering energy) of the liquid means that less molecules are able to escape. i.e. as vaporisation happens, the temperature of the liquid falls causing less vaporisation.
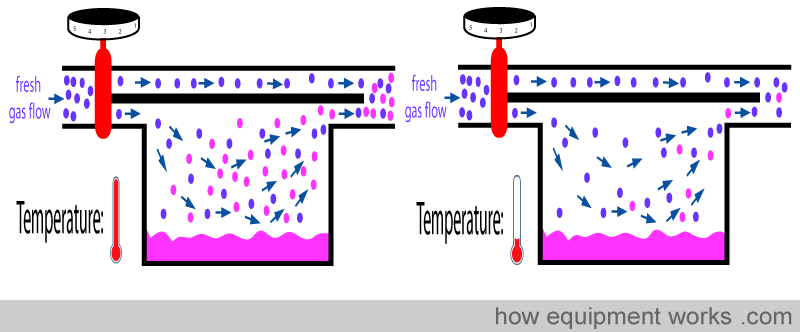
The less vaporisation then will decrease the concentration of anaesthetic delivered by the vaporiser. I.e. It will deliver an anaesthetic concentration below the setting you dialed.
There are two common solutions to this problem.
One is that we can give heat to the liquid to minimise the temperature drop.
The other is to increase the flow of fresh gas into the vaporising chamber to compensate for the reduced vaporisation efficiency of the cold fluid.
‘GIVING HEAT’
In most vaporisers, we don’t actually give heat “actively”. That is, we don’t electrically heat it (complicated and needs a power supply) and nor do we light a fire under it (absolutely dangerous). Instead, we make it easy for the vaporiser to use heat from the surrounding air.
The vaporising chamber is generally surrounded by a lot of metal.
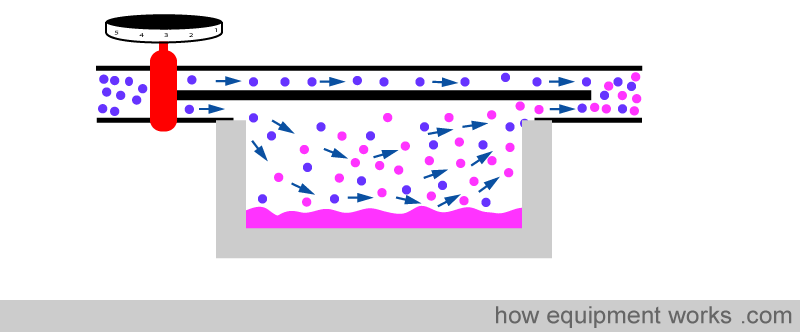
The metal helps to minimise the temperature drop by two ways. Firstly metal is a very good conductor of heat and therefore is able to efficiently transfer heat from the surrounding air into the anesthetic agent.
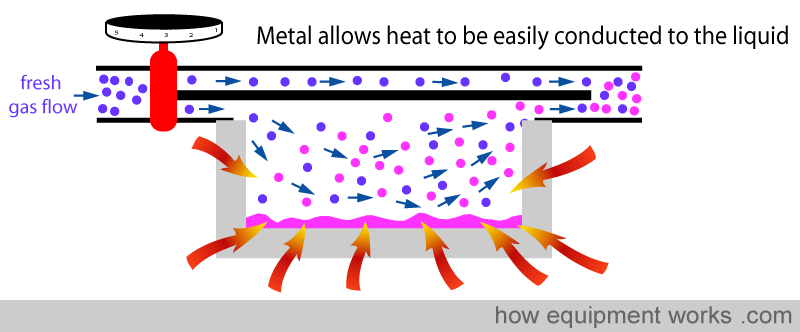
Secondly, metal acts like a ‘heat store’. It ‘absorbs’ heat (green arrows) till its temperature equals the temperature of the surrounding air.
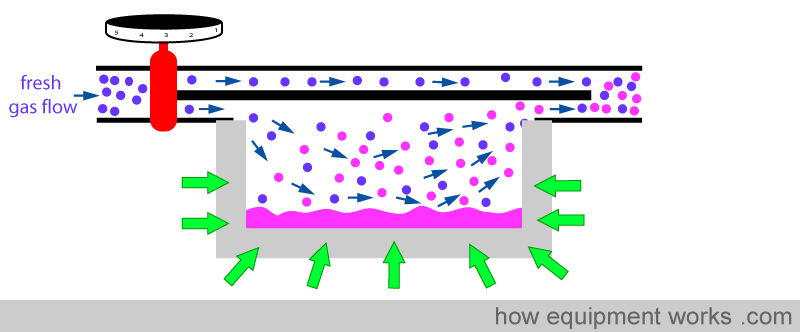
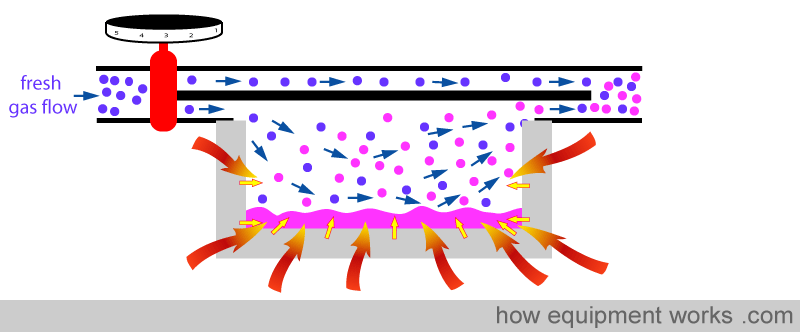
When the anaesthetic agent starts to cool, the metal now ‘donates’ heat ( yellow arrows ) , helping to minimise the temperature drop.
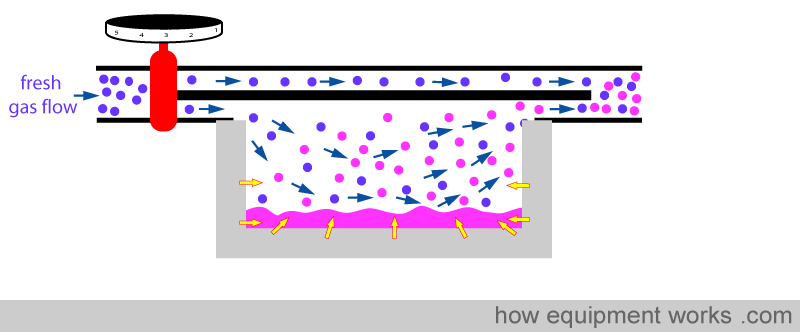
However, the metal casing cannot give up heat indefinitely and after sometime, its temperature also drops.
In between your anaesthetic, when you turn the vaporiser off and have coffee before your next case, the metal will continue to “absorb” heat from the surroundings and its temperature will rise, ready to donate heat when you turn the vaporiser on again.
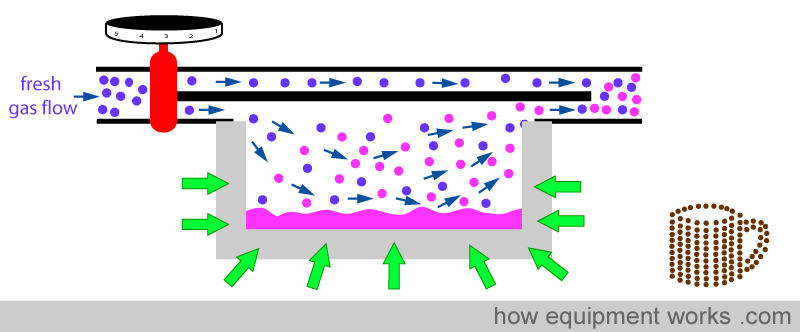
So in summary, the metal provides heat to minimise the temperature drop by two ways. One way is by ‘donating’ heat to the fluid (yellow arrows) and the other way is by conducting heat (red arrows) from the surrounding air.
GIVING MORE FLOW
When the temperature of the liquid agent drops, we have seen that the output concentration of the vaporiser drops. A way of compensating for that problem is to increase the flow of gas via the vaporising chamber (altering the splitting ratio). One could manually do this by measuring the temperature of the liquid with a thermometer and increasing the dial setting according to some kind of reference chart. This would be quite tedious as you would have to do it all the time. Modern vaporisers have removed the hard work. When the liquid drops its temperature, the flow of gas through the vaporising chamber is automatically increased without you having to turn the dial.
This is accomplished by an automatic temperature compensating valve that influences how much flow goes via the vaporising chamber.
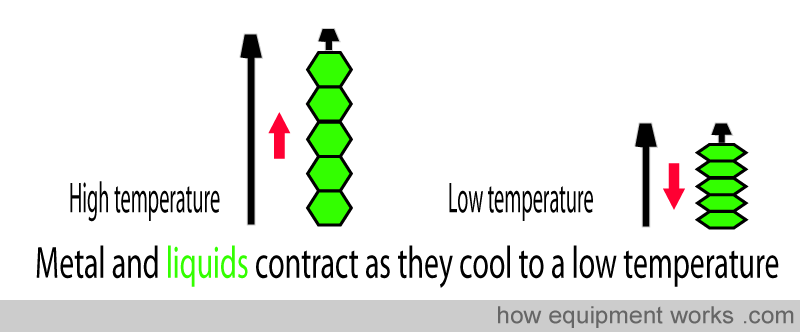
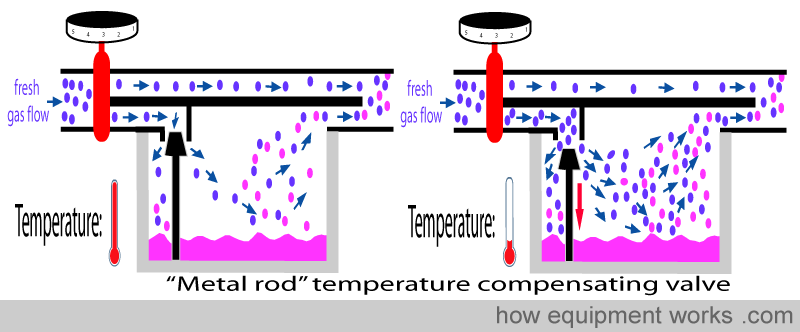
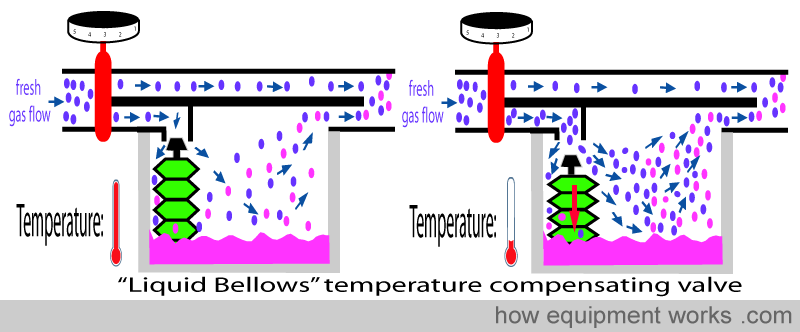
The automatic temperature compensating valve uses the physical property that substances (e.g. metals and liquids ) become smaller when the temperature lowers. A metal rod (shown in black below) shortens as the temperature drops. Similarly, a liquid filled in collapsing bellows (shown in green below) becomes smaller in volume when cooled to a lower temperature.
This property is used in the design of automatic temperature compensating valves in vaporisers. In the design that uses a metal rod, the rod offers some resistance to flow into the vaporising chamber. As the vaporiser cools, the rod becomes shorter, making the valve move away from the opening. This reduces the resistance to flow and thus more flow occurs into the vaporising chamber.
Some vaporisers use the expansion or contraction property of a special liquid inside bellows (shown in green) to control the valve. As the temperature falls, the liquid in the bellows contracts into a smaller volume. This makes the bellows shrink, pulling the valve away and thereby increase flow.
Another method uses a “bi metallic” strip. Different metals expand and contract to differing extents when exposed to temperature changes. In the example below, the “green” metal expands and contracts more than the “red” metal.
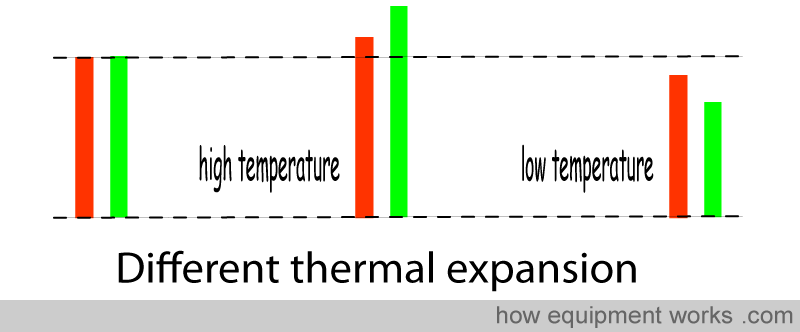
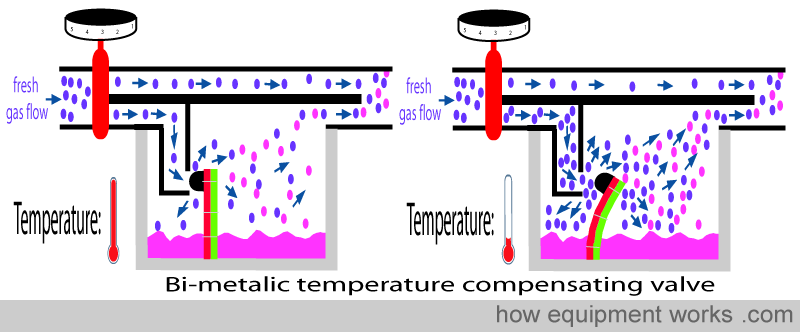
In a bimetallic strip, two metals with very different degrees of thermal expansion ( “different coefficients of thermal expansion” ) are fixed together. In the example below, when the temperature drops, the “green” metal contracts much more than the “red” metal. Because they are fixed together, they cannot contract independently, like in the diagram above. Instead, the “green” metal “tries” to drag the “red” metal and causes the bimetallic strip to bend.

In the vaporiser, the bimetallic strip is fixed in such a way that it offers a resistance to flow entering the vaporising chamber. When the temperature of the vaporising chamber drops, the bimetallic bends and moves away. This reduces the resistance to flow and thus more flow occurs into the vaporising chamber.
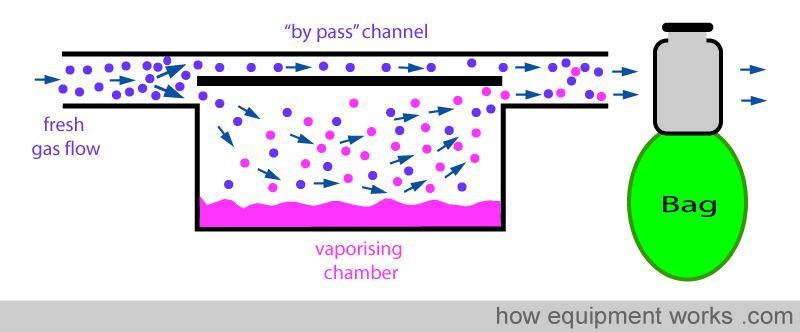
THE “PUMPING EFFECT”
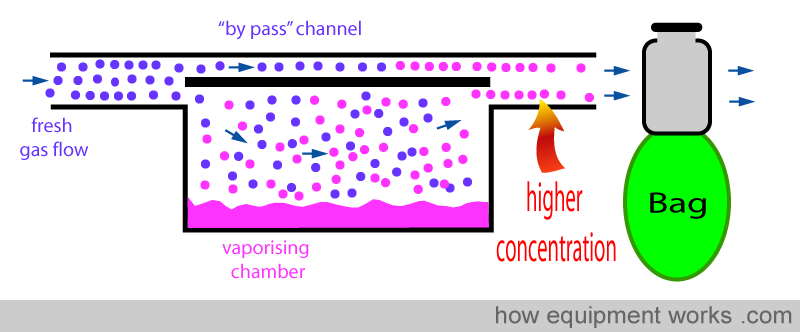
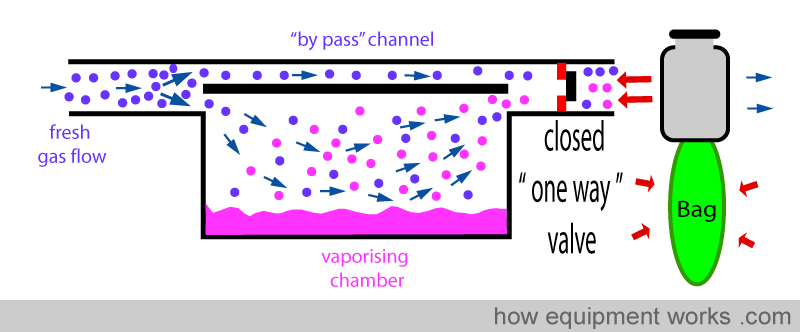
Positive pressure ventilation result in intermittent pressure changes. During the positive pressure, there is a pressure rise and during expiration, there is a sharp drop in pressure. These pressure changes can be transmitted back into the vaporiser and can affect the concentration of anaesthetic agent delivered. The effect of changing pressure affecting the output of the vaporiser is called the “pumping effect”. In this section, this effect and the methods used by vaporiser designers to prevent it from happening are explained. Below is shown a basic vaporiser and beyond it a bag to represent positive pressure ventilation.
When the bag is squeezed (positive pressure ventilation), pressure is transmitted back into the vaporiser as shown below. This “back pressure” is transmitted to both, the “by pass” channel and also to the vaporising chamber. This “back pressure” opposes the flow of the fresh gas in both the “by pass” channel and the vaporising chamber. The fresh gas entering the vaporiser tries to move forward and gets compressed both in the ‘by pass’ channel and the vaporising chamber. However, the vaporising chamber volume is much larger than the ‘by pass’ channel volume, and thus, more fresh gas gets compressed into it than into the ‘by pass’ channel.
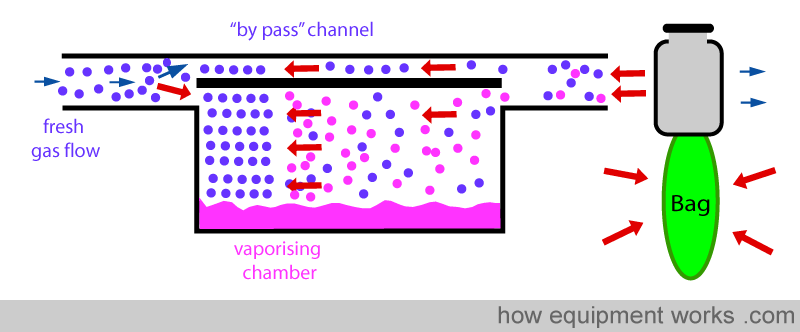
This extra fresh gas that enters the vaporising chamber collects anaesthetic vapor.
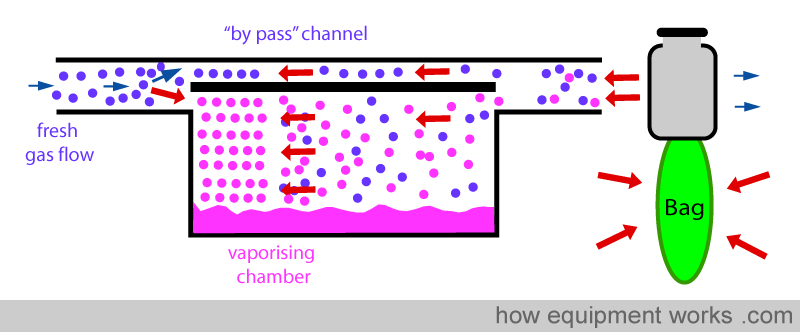
Now see what happens when the positive pressure is suddenly released (expiration). The previously compressed gases now suddenly expands in all directions.
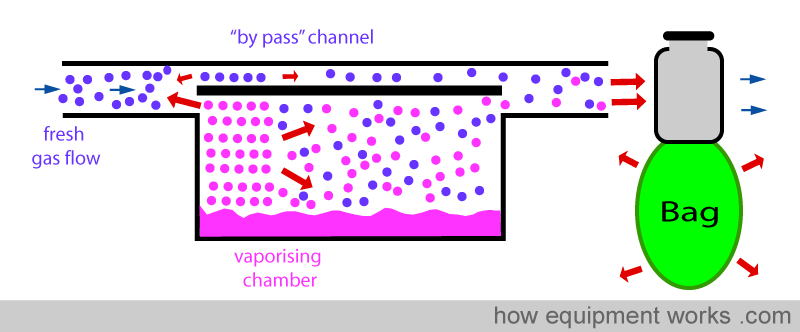
Some of the rapidly expanding gas (containing vapor) enter the inlet of the vaporiser and cross over into the ‘by pass’ channel as shown below.
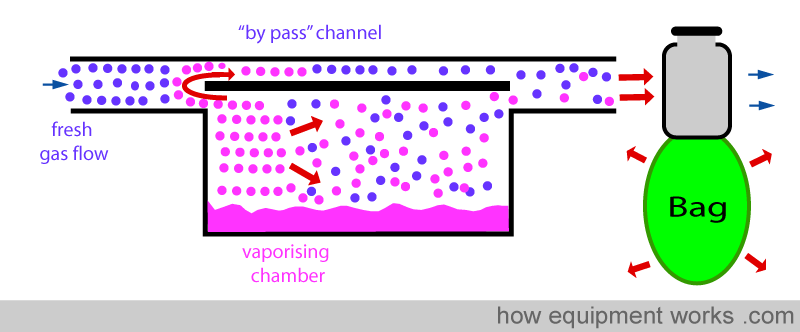
Normally, a vaporiser ‘by pass’ channel does not have vapor. So this vapor due the ‘pumping effect’ is additional. When this ‘by pass’ vapor flows across to the exit of the vaporiser, it meets the vapor from the vaporising chamber. The addition of the ‘by pass’ vapor to the vapor from the vaporising chamber raises the final concentration of anaesthetic delivered. i.e. The ‘pumping effect’ increases the delivered concentration of anaesthetic agent.
Vaporiser designers have various tricks to reduce the ‘pumping effect’ and some of these are discussed below:
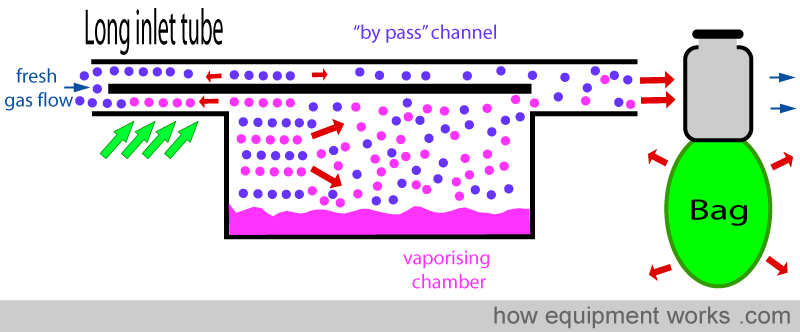
LONG INLET TUBE
The vaporiser inlet tube can be made longer. When the ‘back pressure’ is suddenly released during expiration, as discussed before, the extra gas in the vaporising chamber will suddenly expand. However, thanks to the long inlet tubing, the extra gas containing vapor expands into the long inlet tube and doesn’t reach the ‘by pass’ channel.
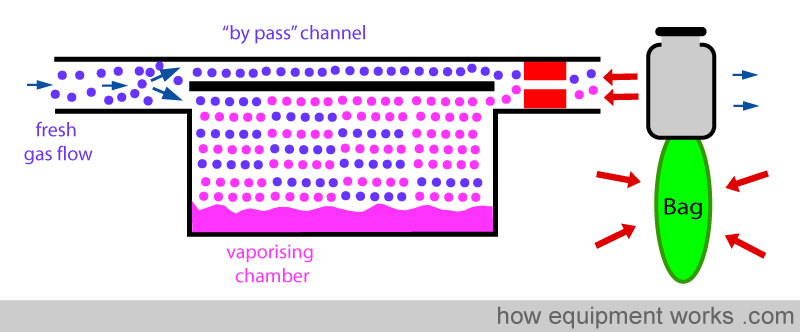
INCREASED RESISTANCE
The vaporiser can be designed to have a high internal resistance to flow. This high resistance “resists” changes to flow caused by the intermittent ‘back pressure’ of positive pressure ventilation.
ONE WAY VALVE
A ‘one way’ valve (also called unidirectional valve) can be put between the vaporiser outlet and the ventilator / breathing system. On way valves allow flow in one direction, but not in the other. In the diagram below, the one way valve is allowing gases to flow forwards.
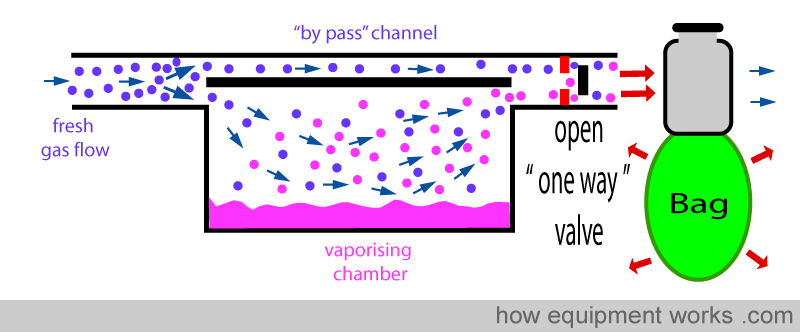
However, this valve prevents flow from occurring in the reverse direction. This reduces the transmission of ‘back pressure’ to the vaporiser.

Everyone wants to be happy. However, according to psychologists, many of us think in certain “unhelpful” ways which can prevent us from being happy. The free website at the link below describes the concept of “Happy Thinking”, which is about using simple methods to change such ways of thinking. You are welcome to visit it and find happiness by changing how you think. The website is completely free.

Desflurane Vaporizer
Before we proceed to talk about the desflurane vaporizer, we need to understand what vapor pressure is.
VAPOR PRESSURE (also called SATURATION VAPOR PRESSURE)
The process of evaporation in a closed container will proceed until there are as many molecules returning to the liquid as there are escaping (equilibrium). At this point the vapor is said to be saturated, and the pressure exerted by the vapor (usually expressed in mmHg) is called the saturated vapor pressure.
Since the molecules move faster (more kinetic energy) at higher temperature, more molecules can escape the surface and the saturated vapor pressure is correspondingly higher. i.e. higher the temperature, higher is the saturated vapor pressure.
The temperature at which the vapor pressure is equal to the atmospheric pressure is called the boiling point.
Desflurane has a very low boiling point (about 23 degrees Centigrade) and even at room temperature, has an high vapor pressure.
Also, for small changes in temperature, the vapor pressure of desflurane changes quite dramatically. I.e. desflurane is said to have a very steep “Vapor Pressure versus Temperature curve”.
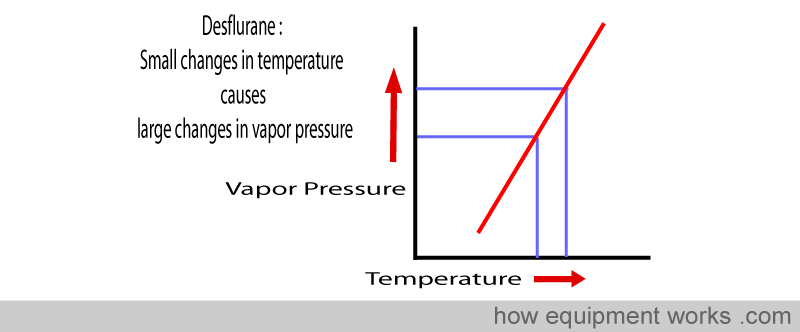
These physical properties of desflurane creates a big headache for vaporiser designers.
An operating room temperature is not perfectly constant. It keeps changing slightly depending on various factors including the number of medical students (young body heat) watching the surgery. These changes in operating room temperature then change the temperature of vaporisers present in that room. As discussed elsewhere, the standard vaporisers try to resist changes in temperature (e.g. by having thick metal construction). However, these mechanisms are not perfect and in practice small changes in vaporiser temperature still occur. This is not a big problem with anaesthetic agents such as Isoflurane or Sevoflurane which have a relatively less steep “Vapor Pressure versus Temperature curves”. In them, small temperature changes will lead to only small changes in vapor pressure and this can be compensated by mechanisms such a the bimetallic strip. With Desflurane, with its steep “Vapor Pressure versus Temperature curve”, even these small temperature changes can cause large changes in vapor pressure which cannot be compensated for with simple devices such a bimetallic strip. So a whole new vaporiser design had to be made.
The solution chosen for the problem is to have a vaporiser that heats the Desflurane to a very precisely controlled temperature that is not affected by changes in room temperature. The heated vapor is then “injected” into the fresh gas flow.
You will recall that “standard” vaporisers work by splitting the fresh gas flow into two pathways, one going through the vaporising chamber and picking up anaesthetic agent and the other “by passes” the chamber and thus has no anaesthetic. The two streams then mix at the end of the vaporiser to give the final concentration of anaesthetic.

The desflurane vaporiser works differently. It “injects” the anesthetic agent directly into the fresh gas flow. In this method, the fresh gas flow coming from the flow meters does not split into two streams. There is only one stream for the fresh gas flow, and into this stream, the anaesthetic agent is directly injected.
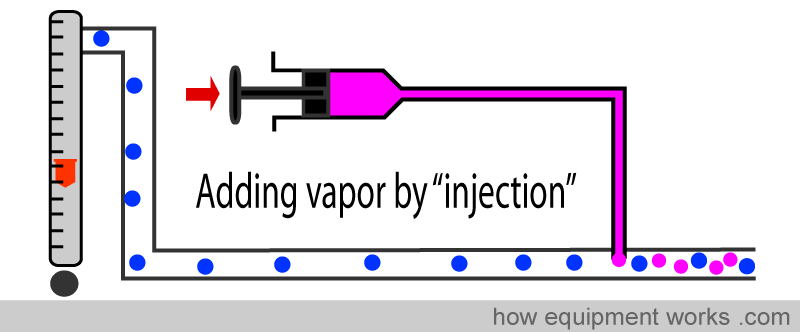
However, the design is more complicated than the simple syringe system shown above. The system is more complex; but don’t worry, we will go through each part of it slowly.
There is a tank (sump) which contains desflurane which is electrically heated to a highly controlled constant temperature (approximately 40 degrees C). Because of the heat, the liquid Desflurane becomes gaseous Desflurane at a pressure of about two atmospheres (about 1500 mmHg or 200 kPa). This Desflurane gas is injected into the fresh gas flow.
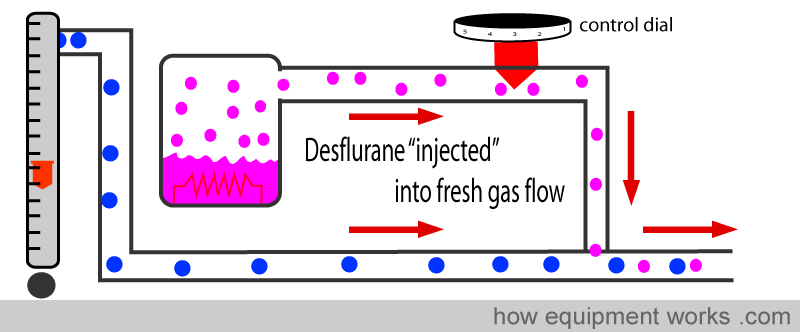
The amount of Desflurane concentration in the fresh gas is controlled by the dial setting set by you. The dial moves a valve which varies the resistance to Desflurane flow from the tank to the fresh gas.
If you want a higher concentration of desflurane, the valve attached to the dial reduces the resistance to flow of desflurane and more of it gets injected into the fresh gas.
Conversely, if you want a lower concentration of desflurane, the valve attached to the dial increases the resistance to flow of desflurane and less of it gets injected into the fresh gas.
The rate of desflurane gas injection must be adjusted to match the fresh gas flow going through the vaporiser.
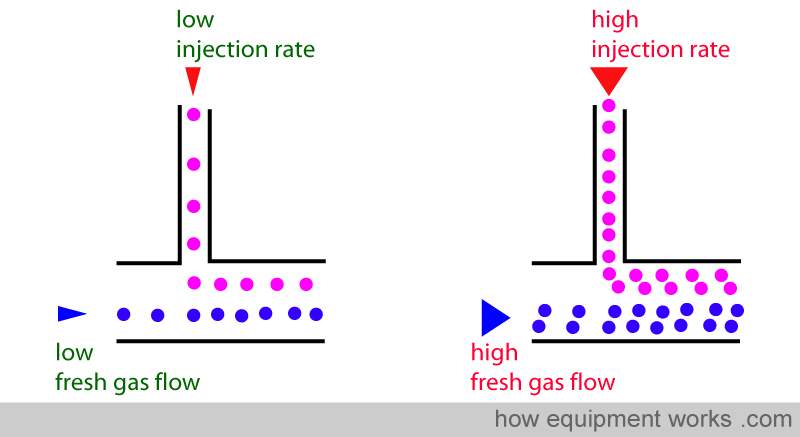
If you increased the fresh gas flow, but didn’t increase the injection rate, the emerging mixture will now be inaccurate, the concentration being lower than before.
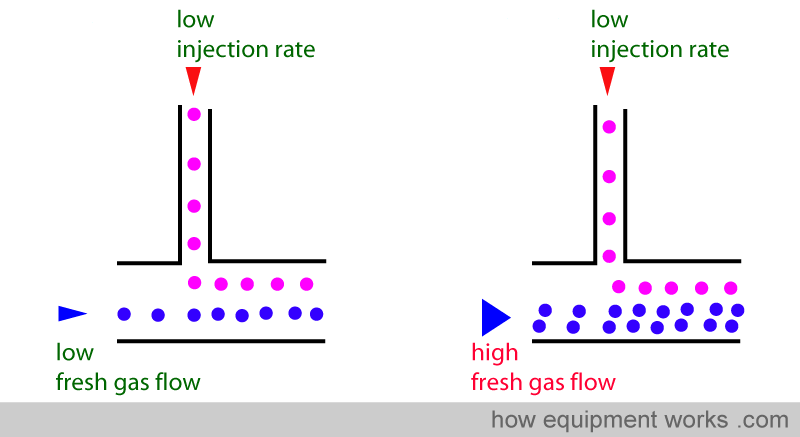
Similarly, if you decreased the fresh gas flow, but didn’t decrease the injection rate, the emerging mixture again will be inaccurate. This time, there will be relatively more anaesthetic agent , making the mixture higher than intended.
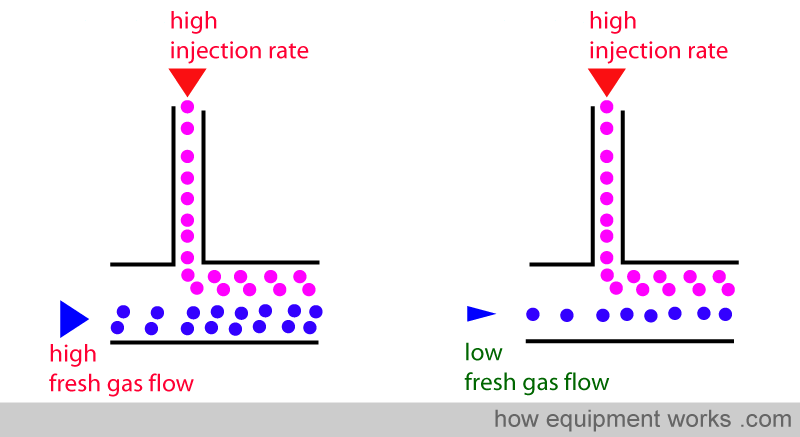
One solution would be for you to manually adjust the dial setting to match the fresh gas flow. For low flows, you will have to reduce the dial setting to reduce the rate of Desflurane injection, and for high fresh gas flows, you will need to do the opposite. This would be really tedious in our modern times. Fortunately, the Desflurane vaporiser automatically adjusts the rate of injection of desflurane to match the flow rate, and thus keeps the delivered concentration constant.
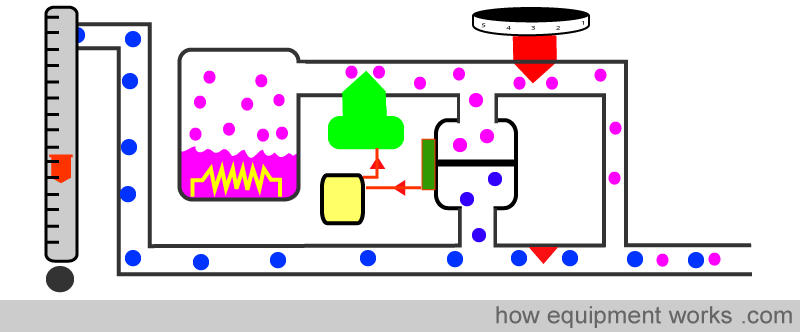
We are now ready to discuss the workings of the Desflurane vaporiser. You will need to refer to the numbers on the diagram under the description.
Your flow meters deliver the fresh gas flow (1). The fresh gas travels through the pipe (2). Note that, unlike other vaporisers, none of the fresh gas goes to the vaporising chamber (4).
The vaporising chamber is electrically heated (3). Using sensors for feedback, the temperature is kept very constant. The heating causes the Desflurane to become a gas under pressure (4) and this travels down pipe (5).
The flow of Desflurane is resisted by two valves (6,13). Valve (6) is the valve that you control when you set the dial to a particular concentration. When you increase the concentration setting, the valve (6) opens a bit and lowers the resistance, allowing more Desflurane to flow through. Valve (13) is an electronically controlled valve. Computer (12), the vaporiser’s “brain”, is able to also alter the flow of Desflurane by controlling the valve (13). i.e. both you and the computer can adjust the desflurane injection rate. The Desflurane then goes via pipe (7) and meets the fresh gas at (8). The Desflurane mixes with the fresh gas (8) and a final concentration emerges from the exit of the vaporiser (9).
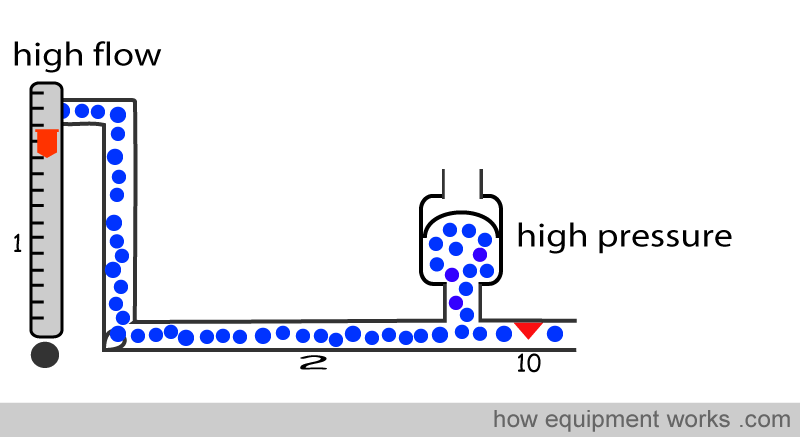
Now we can discuss how the vaporiser, to keep the output concentration accurate, adjusts the Desflurane flow when the fresh gas flow changes. As explained before, the fresh gas flows in pipe (2). This pipe has a fixed resistance (10) in its path. For the fresh gas flow to overcome this resistance (10), the pressure in pipe (2) rises. Higher the fresh gas flow in pipe (2), higher will be the pressure rise in pipe (2) since more flow has to occur through the same fixed resistance (10).
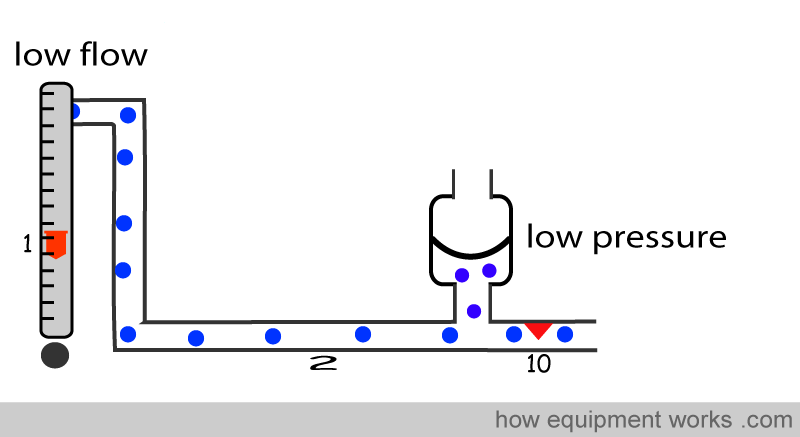
Similarly, when the fresh gas flow is decreased, the lesser flow will find it easier to go through the fixed resistance and the pressure in pipe (2) drops. It is important to remember that the pressure in pipe (2) is proportional to the fresh gas flow going through it. Higher the flow, higher is the pressure in pipe (2). Lower the flow, lower is the pressure.
Device (11) is called a “differential pressure transducer”. It has a diaphragm that on one side is exposed to the pressure in pipe (2) carrying fresh gas and the other side of the diaphragm is exposed to the pressure in pipe (5) carrying Desflurane. When the pressure is equal on both sides of the diaphragm, it lies in a neutral position. i.e. pressure P 1 equals pressure P 2.
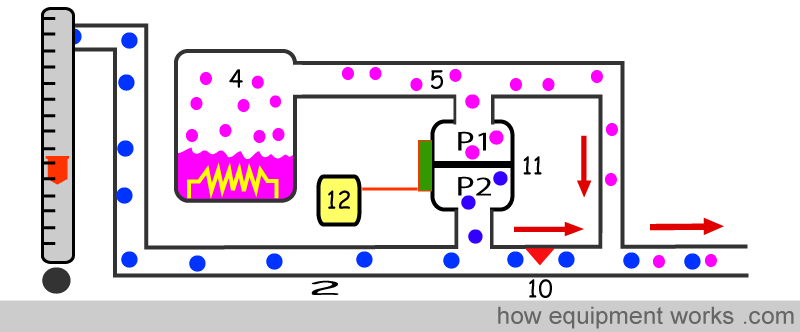
If one side of the diaphragm is at a higher pressure than the other side, the pressure difference makes the diaphragm move. In this way, the differential pressure transducer (11) is able to measure the pressure difference between the fresh gas flow pipe (2) and the Desflurane flow pipe (5). It continuously keeps the computer (12) informed about pressure difference information.
Now let us see how the vaporiser copes when the fresh gas flow is increased. The fresh gas flow has been increased by you (1). Increased fresh gas flow flows through pipe (2) and meets fixed resistance (10). The increased flow through the fixed resistance (10) makes the pressure in the pipe (2) to rise and this pressure is experienced by the differential pressure transducer (11). Since the desflurane pressure in pipe (5) is now lower than the fresh gas pressure in pipe (2), the diaphragm in the differential pressure transducer (11) moves and a signal about the pressure difference is sent to the computer (12).
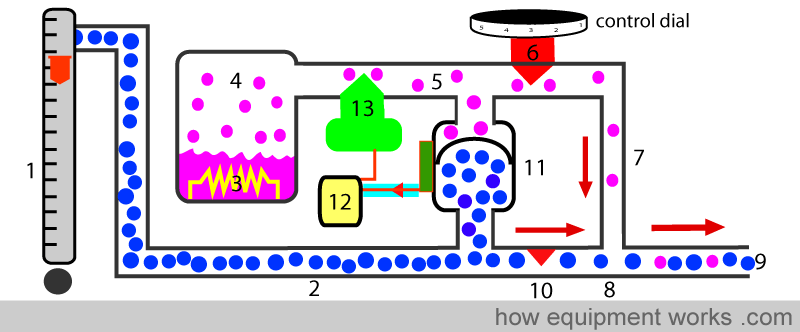
The computer (12), acts on the information provided by the differential pressure transducer. It proceeds to increase the flow of desflurane to inject into the increased fresh gas flow. It commands the electronically controlled valve (13) to reduce the resistance to flow. As the valve (13) opens up and lowers the resistance, the Desflurane flow increases.
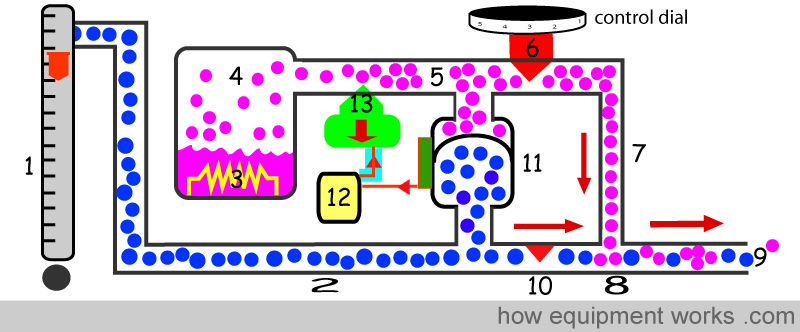
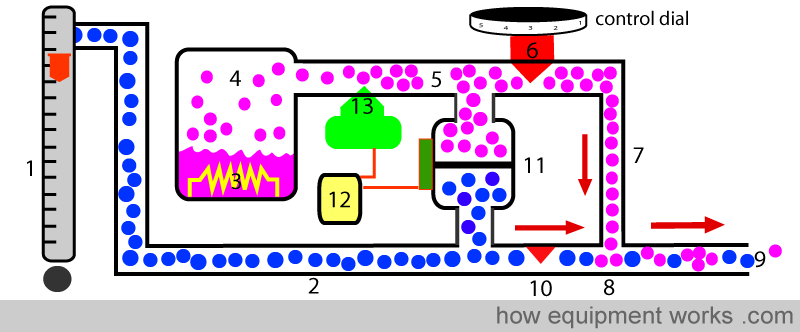
The increased flow of Desflurane causes the pressure in pipe (5) to rise. This pressure rise pushes the diaphragm of the differential transducer back to its neutral position (11). The differential transducer (11) informs the computer (12) that the diaphragm is in the neutral position. The computer (12) is now happy that it has increased the flow of desflurane sufficiently to match the increased fresh gas flow rate and it therefore stops further opening of valve (13). Since the two flows are matched, the output concentration (9) does not change despite the increased fresh gas flow. If you again change the fresh gas flow rate, the system will again adjust the desflurane injection rate.
Interlock Mechanism
Many anesthetic machines have more than one vaporiser attached so that one has a choice of inhalational agents to use. However, it is important that only one vaporiser be used at a given time to avoid overdose with different vapors going into the patient simultaneously. There are many different safety mechanisms available which prevents more than one vaporiser to be used simultaneously. I describe one such system below. Please note that your anesthesia machine may use a different system.
In this system, each vaporiser has two pins protruding out. When the vaporiser is in use, the pins protrude outwards. When the vaporiser is turned off, the pins retract back to where they were.
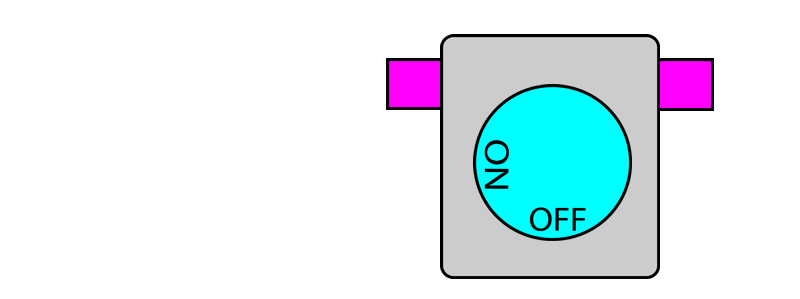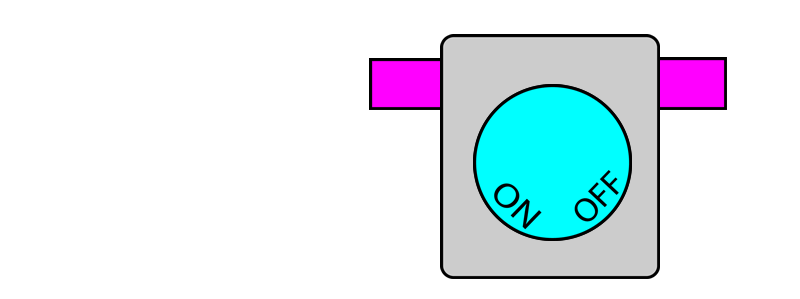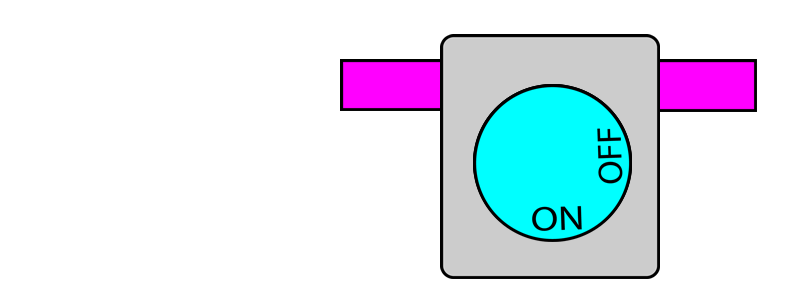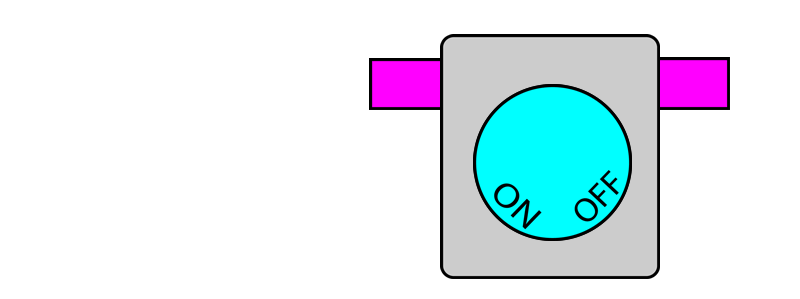
On the other hand, if any of the pins are pushed in (i.e. by another vaporiser) this locks the vaporiser dial in the OFF position. When the pin is no longer pushed in, the dial once again becomes unlocked and can be turned.
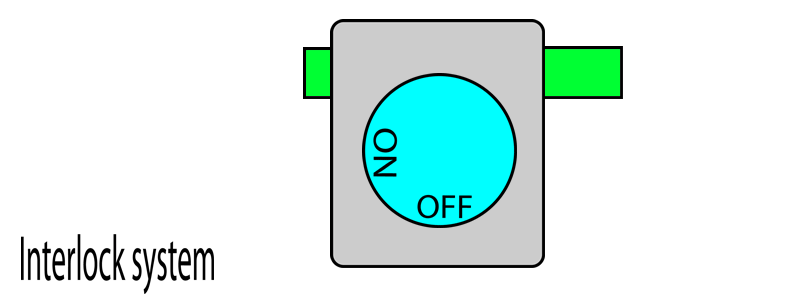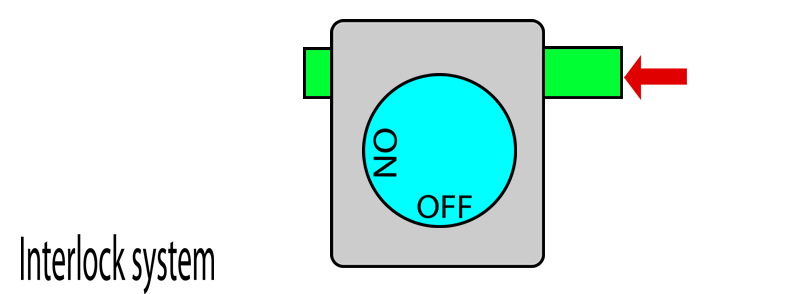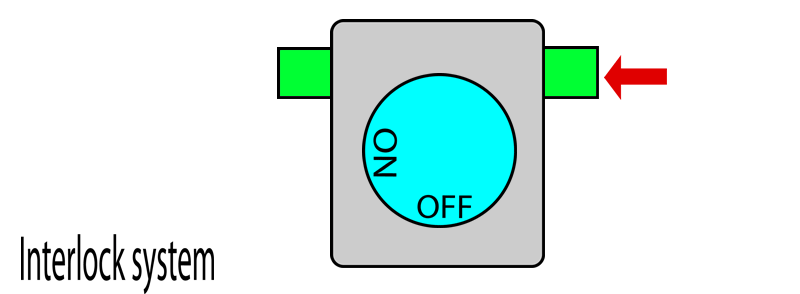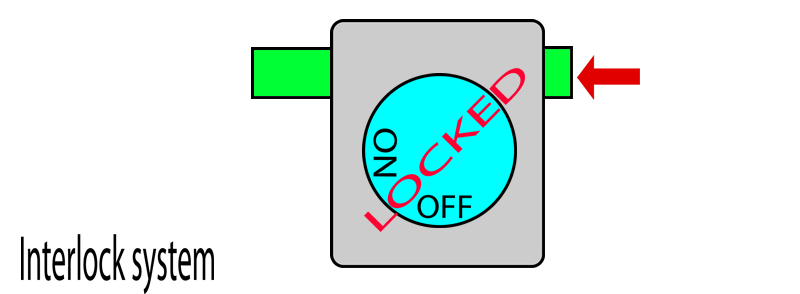
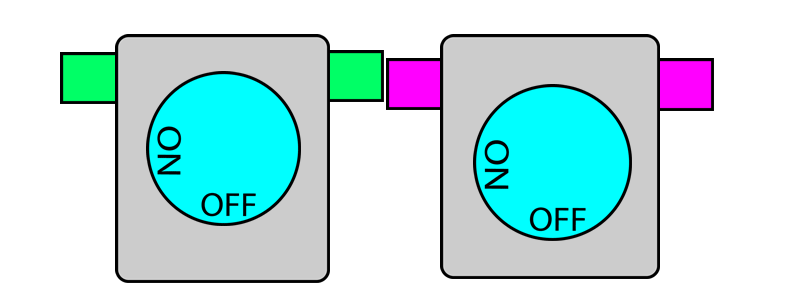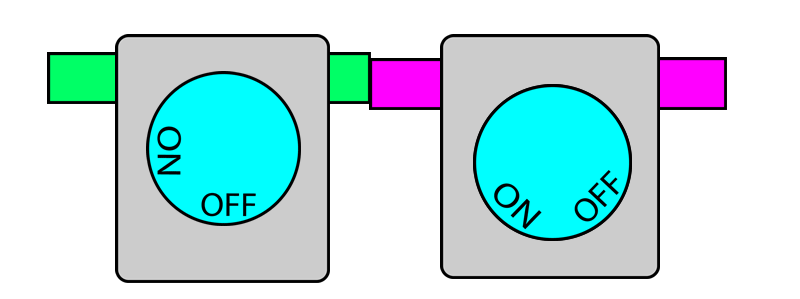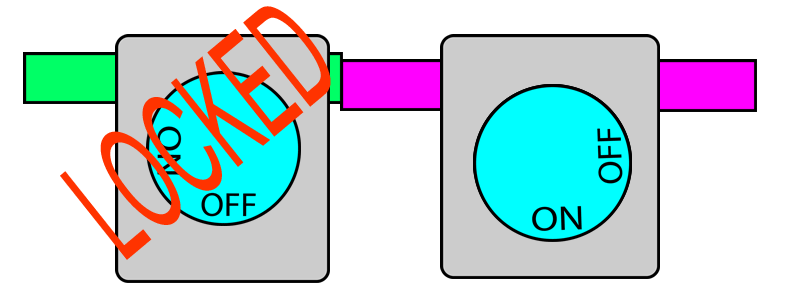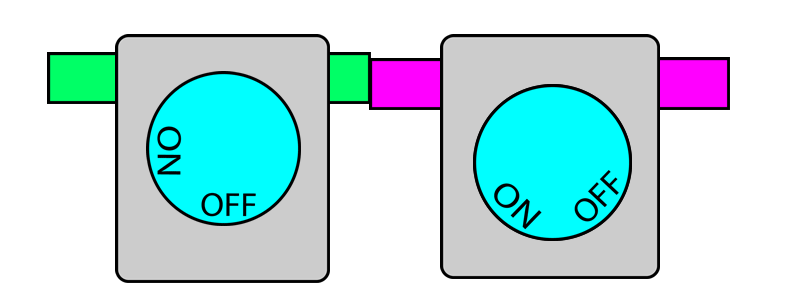
When you put two vaporisers together, their pins touch.
When one vaporiser is turned on, it protrudes its pins which then pushes in the pins of adjacent vaporisers and locks them. When this vaporiser is turned off, its pins retract and releases the pins on the adjacent vaporisers and thereby unlocks them. In this way, only one dial can be turned on at a given time.
Below is a photograph of an interlock mechanism.

Agent Specific Fillers
It is important to fill the correct agent into the correct vaporiser. If a wrong agent is filled into a vaporiser, you will be giving the wrong drug, and worse, since vaporiser designs for different agents vary, you may seriously overdose your patient.
Early vaporisers had simply a funnel into which you could pour virtually anything by mistake (including coffee).
Modern vaporisers have special filling systems specific for each anaesthetic agent to prevent inadvertent filling with an wrong agent. Think of it as a “lock and key” system, i.e. a particular key will fit only a specific lock.
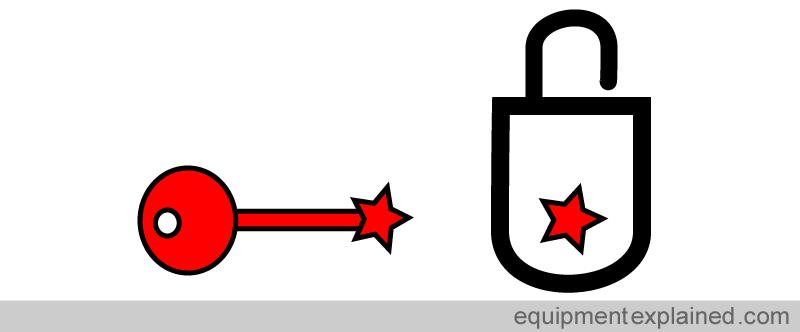
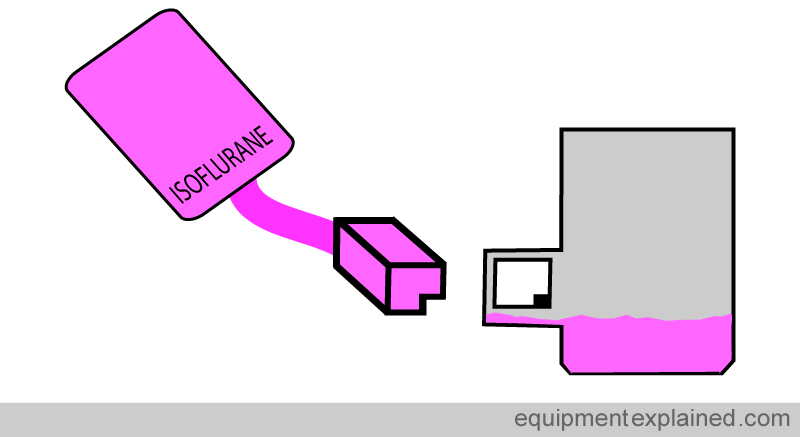
There are various systems in use. In the system below, the Isoflurane filler (key) has a notch in a corner. This fits perfectly with the filling hole in the Isoflurane vaporiser. The filling hole has pin at the corner over which the notch of the Isoflurane filler key can pass over.
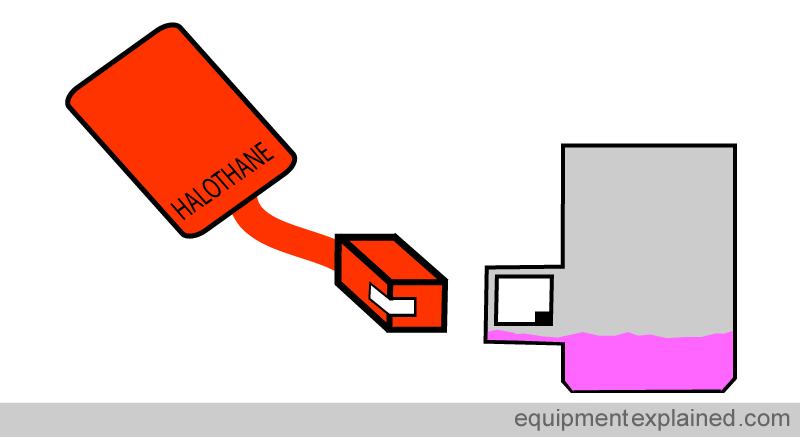
A different anaesthetic agent such as Halothane (not commonly used anymore) has a different filling key. In this case, the key has a notch at the side instead of at the corner. So the Halothane filler key will not fit into the Isoflurane vaporiser filling hole.
The system described above is only one type of agent specific filling system. There are others that are there and depend on the manufacturers and the country you work in.
In addition to the physical shapes being different, the key fillers are also color coded (purple for Isoflurane, yellow for Sevoflurane, blue for desflurane).
Here are some actual images of an filler in use. However, please note that the system used in your country / hospital may be different from what is shown.
Isoflurane Filler
The Isoflurane bottle has notches in them arranged in a way that is specific for Isoflurane. The Isoflurane key filler has specific corresponding cuts where the notches of the bottle will fit. This makes sure that you cannot fix the wrong filler key into the wrong bottle.

The notches on the bottle fit perfectly into the key filler.

The correct key filler is on the correct bottle and is ready.

Note the corner notch in the vaporiser end of the Isoflurane key filler.

The corner notch in the Isoflurane key filler aligns with the corner notch of the Isoflurane vaporiser.

Filling with the correct agent.

We have now reached the end of our discussion on anaesthesia vaporisers. I hope it has given you a good introduction to the subject and will help you when you read further on this topic. Bye and see you soon at another topic !

Hello! My name is Pras and I am the author of this website that you are now reading. I have made this website completely free to access so that people from all over the world can benefit from it.
If you can afford it, I would be very grateful if you would consider making a single donation of one dollar (or the equivalent in your currency) to help cover the expenses needed to run this website (e.g. for special software and computers). For this website to survive, donations are desperately needed. Sadly, without donations, this website may have to be closed down.
Unfortunately, perhaps because many people think that someone else will donate, this website gets only very few donations. If you are able to, please consider making a single donation equivalent to one dollar. With support from people like you, I am sure that this educational website will continue to survive and grow.


